|
阅读:2388 ,发布于2023/11/15 10:43
A 65 page guide on the practical aspects of performing SPPS
What is this guide?
Written through more than 60 years of combined experienced in making peptides,
this is a 65 page practical guide on SPPS.
The purpose of this guide is two-fold. First, a brief introduction on
the development and most common applications of solid-phase peptide synthesis
will enable the user to best apply the two most widely-used synthetic
strategies – Boc/Benzyl and Fmoc/tButyl chemistries – to his/her projects.
Second, a detailed description of peptide synthesis, cleavage, and purification
in the experimental section is given with ‘helpful hints’ so that newcomers to
peptide science will have easy access and avoid some of the obstacles which
often lead to expensive mistakes and/or poor synthesis yields
Introduction
Solid phase synthesis is
a process by which chemical transformations can be carried out on solid support
in order to prepare a wide range of synthetic compounds. This idea was first
developed by Bruce Merrifield to synthesize polypeptides and earned him the
Nobel Prize in 1984. Solid phase chemistry offers many advantages over
conventional synthesis in terms of efficiency as well as convenient work-up and
purification procedures. In solution phase peptide synthesis, particularly in
longer sequences, the repetition of coupling and deprotection cycles can become
very labor intensive and require the isolation of all peptide intermediates.
I. A Brief Historical
Perspective The chemistry of peptide synthesis – first developed in the
early 1900’s by Emil Fischer – arguably marks the birth of organic synthesis as
we know it today. Whereas the lion share of organic synthesis continues to be
performed by solution-phase methods, i.e., with each independent chemical
reaction followed by a purification step and characterization of the resulting
synthetic intermediate, two peculiarities of peptide chemistry spurred the
development of a more efficient synthetic strategy. In contrast to most total
synthesis efforts, the synthesis of peptides – at least until the final
deprotection step – is an iterative process, with alpha-amino (alphaN)
deprotection and amide couplings performed in succession until the desired
full-length target peptide is obtained. In addition, most peptides of
biological interest are grossly insoluble in most organic solvents irrespective
of side-chain protection tactics. The net result of these two characteristic
features of peptides was that the first sixty-odd years of peptide synthesis
forged little ground until the landmark work in the late 1950’s of R.B.
Merrifield at Rockefeller University.
During the course of his
Ph.D. work, Merrifield (a biochemist) proposed an entirely new paradigm in
organic synthesis. As is often the case when an outsider looks into an insular
field and pursues a tack that is anathema to the existing experts in the field,
Merrifield’s idea of performing all synthetic manipulations using the
C-terminus of the target peptide linked to an insoluble solid support was met
with much criticism. However, it was not long before the chemistry of
solid-phase peptide synthesis (SPPS) was honed to a point where traditional
solution-phase methodologies were no match with regard to speed and
versatility. The original “Merrifield” version of SPPS – more accurately
referred to as Boc/Benzyl chemistry – was roughly finalized in the late 1960s,
and employs a graduated acid lability system for manipulation of all
protecting groups (Scheme 1). In this strategy, the alpha-amino
t-butoxycarbonyl (Boc) protection is removed with TFA, while side-chain
protections and the peptide- resin anchorage (the linker) require much harsher
acidic conditions for cleavage. This final step is accomplished using liquid
HF, a much stronger acid than TFA (acidity functions of -11 and 0.1,
respectively).
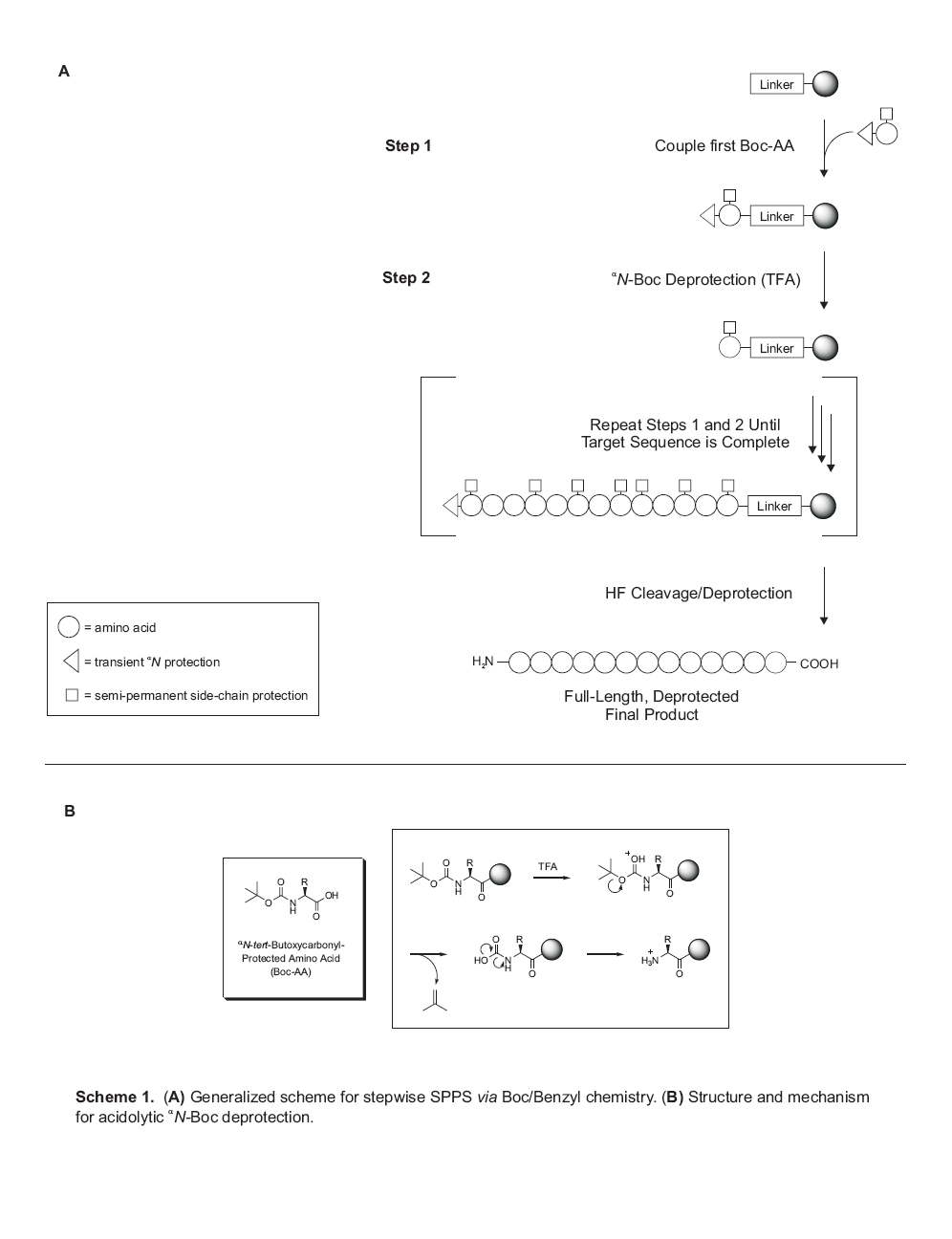
Scheme 1 depicts the manner in which Boc/Benzyl SPPS simplifies all of
the reactions involved in peptide synthesis by allowing for purification via
filtration, so that excess reagents can be employed and removed by simple
washing. It is important to emphasize that the chemistry of SPPS is not
fundamentally different from that used in solution-phase peptide synthesis. The
sole chemical distinction between solution- and solid-phase peptide synthesis
is that the C-terminal protecting group in the latter is rendered
insoluble by virtue of its incorporation into a polymer. This difference
notwithstanding, all side-chain and alphaN protecting groups, as well as
coupling chemistries, employed in solid-phase synthesis have been successfully
applied in solution-phase synthesis, and vice-versa, with few exceptions.
During the 1970s several groups were actively developing milder methods
for SPPS that avoided the use of liquid HF as for the final
deprotection/cleavage reagent. While a variety of milder graduated acid
lability systems were devised, the method that rose to general applicability
was the orthogonal system of Fmoc/tBu chemistry. This strategy – developed by
R.C. Sheppard at Cambridge University – differs from Boc/Benzyl chemistry in
that the side-chain and alphaN protecting groups are removed under conditions
that leave the other class entirely intact. In Fmoc/tBu chemistry, a mild base
– usually piperidine (pKa = 11.1) – is employed for iterative N
9-fluorenylmethoxycarbonyl (Fmoc) deprotection, while global side-chain
deprotection/cleavage is accomplished with TFA (Scheme 2).
It must be emphasized that the more traditional Boc/Benzyl and Fmoc/tBu
chemistries, while differing in chemical minutiae, are fundamentally the same
process. In both cases, the target peptide chain is assembled in a stepwise
fashion from alphaN- and side-chain protected amino acids. In both cases, the
‘transient’ alphaN amino protection is employed solely during chain elongation
(the coupling reaction) and then removed for the subsequent coupling reaction.
Lastly, in both cases, the final step – global side-chain deprotection and
cleavage of the peptide-resin anchorage – is accomplished by acidolysis, and
the target peptide isolated by trituration from ether and purified by
reversed-phase high performance liquid chromatography (RP-HPLC).
Download the full 65 page guide here: Download Link
About CSBio: For over 30 years, CSBio, a leading peptide and peptide
synthesizer manufacturing company located in Silicon Valley, California, has
been providing cGMP peptides and automated peptide synthesizers to the global
pharmaceutical community. CSBio’s peptide products and peptide synthesizers can
be found in production laboratories, universities, and pharmaceutical companies
worldwide.
|
 沪公网安备 31011502005552号
沪ICP备13005633号-1
沪公网安备 31011502005552号
沪ICP备13005633号-1